Welcome To CURA DMS
Make your Document Management System easier to Use for the End Users
Simplification of DOCUMENT CONTROL Procedure eases the work of an Employee with digital approach by reducing 70-80% of complex tasks, saving valuable time for the staff.
DMS Solution
DMS will be a Readymade Solution for the Clients as a Centralized Repository for entire document flow at site
Any Changes happening at Manufacturing site which would impact the Product Quality & Patient Safety along with all types of Process / Material / Document / Method Changes would be captured under this Change Management as per Regulatory guidance’s.
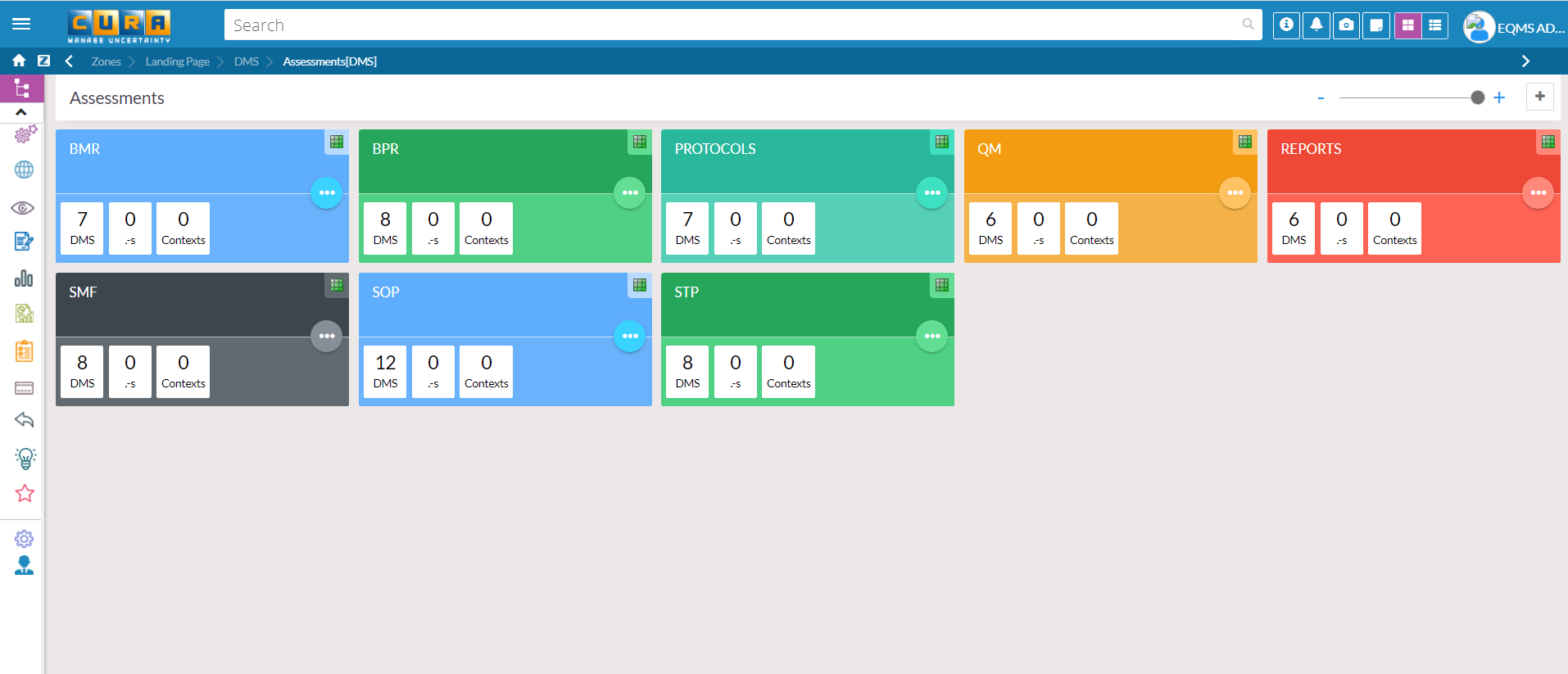
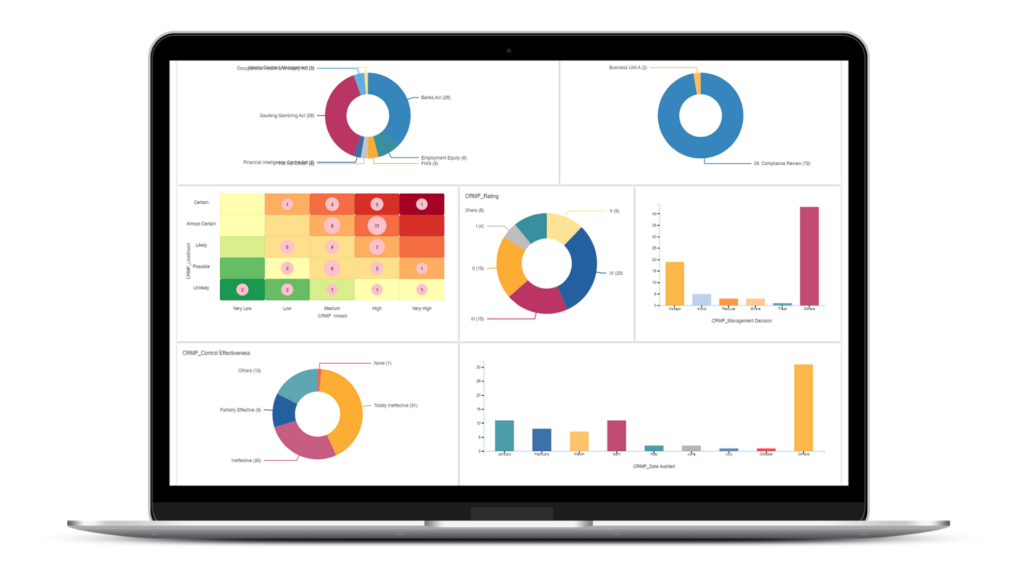
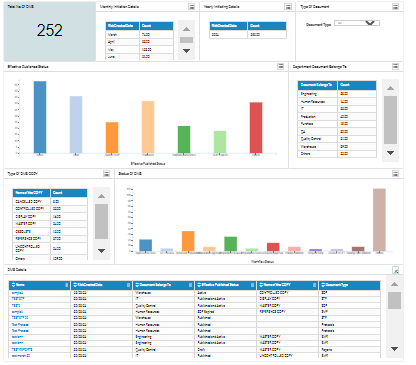
Accurate Data Insights and Reviews for Wholistic out look of DMS
CURA Data Analytics gives a Clear Insight to take the Right Decisions regards to Complete Review by the Management for the Overall Effectiveness of DMS ( Document Management System) at Site.
Features
Easy and Smart Solution for You
Reporting and analytics
To analyze and measure the effectiveness of DMS efforts that organizations can stay on top of regulatory requirements, Procedural Compliance with Document Repository Status like SOPS, STPS, CoAs, Validation Reports, Audit findings, Customer Complaints etc.

Easy Automation
Will help to achieve automated DMS solution.
Data Security
CURA focusses on Data Security. Our DMS utilizes robust actions to safeguard and shield Vulnerable data, Confirming and guaranteeing that the data is safe and secure and compatible conditions to meet compliance for the Customer’s Crucial Data / Information resources.
Easy Configuration
CURA's DMS is highly feasible for integration with other modules and can seamlessly connect with EQMS & SAP / ERP systems for smooth operations. Through API integration, customers can enhance and improve their DMS with Electronic Quality Management Systems ( with CURA's EQMS.
Complying to Global Regulatory Requirements
CURA’s DMS meets the desired regulatory compliance with stringent regulations namely USFDA 21 CFR Part 11 & EU GMP Annex 11etc and ALCOA Principles as well.
User Friendly
The Solution is User friendly and Can be Customizable and Configurable as per Client Needs.

Notifications and Alerts
Proactive Alert Mechanism (Notifications for Approvals / Pop ups for Past Due Actions/ Mail Alerts.
Frequently Ask Questions
EQMS improves quality management by streamlining processes, reducing errors, enhancing traceability, and ensuring consistent application of quality standards across the organization.
While EQMS is often used by large organizations, many solutions are scalable and can be tailored to meet the needs and budget of small and medium-sized businesses.
EQMS automates quality processes by using workflows to handle tasks such as document approvals, CAPA management, audit scheduling, and training tracking, reducing manual effort and the potential for errors.
Data within an CURA EQMS is typically highly secure, with features such as user authentication, role-based access control, data encryption, and regular security audits to protect sensitive quality management information.

